Steve Groothuis — or Groot to everybody else on this planet — Chief Technology Officer of Samtec Microelectronics, recently presented “Packaging Innovations For Medical Wearables” at the Medical Wearables Conference in Santa Clara, California.
Groot’s presentation focused on the specific needs, challenges, and innovations within upcoming medical applications and medical device packaging. We caught up with Groot and asked him about the presentation and its message.
DANNY: Steve, let’s start with a very basic question. What are medical wearables?
GROOT (STEVE GROOTHUIS): “I am Groot.” (Medical Wearable Devices are autonomous, non-invasive devices that perform specific medical functions such as managing treatment of chronic diseases or monitoring vital signs of the human body. A wearable medical device incorporates capabilities such as wireless data transmission real-time feedback, assessment, and so-on.)
DANNY: Groot, what are some of the more commonly-used package types for mobile and wearable devices?
GROOT: “I am Groot.” (The most popular package types are touch screen controllers (Quad Flat No-Lead (QFN) and fine pitch ball grid arrays (FBGA), application processors (Flip Chip Ball Grid Array), and WiFi/Bluetooth (Land Grid Array and Wafer Level Packages. But the most important design element is low profile. Fan-out Wafer Level Packages (FOWLP) have many inherent advantages, including low profile, higher speeds, more I/O’s, higher integration, fewer processing steps, and no substrate required.)
DANNY: What are the most important design attributes of medical wearable devices?
GROOT: “I am Groot.” (Size and weight are obviously huge. Flexibility is also essential. Many of these devices are designed to wrap around your wrist. Examples include the Nike+ FuelBand Packages, Disney’s Magic Band, and IMEC’s flexible wireless ECG sensor.
The following medical measurement technologies are now appearing in more wearable devices:
- ECG/EKG (electrocardiography) = sensors measure bio-potential generated by electrical signals that control expansion and contraction of heart chambers.
- PPG (photoplethysmography) = sensors use a light-based technology to sense rate of blood flow as controlled by heart’s pumping action.
- EEG (Electroencephalography) = sensors monitor electrical stimulations within user’s brain.
- EMG (electromyography) = sensors that record muscle activity.)
DANNY: What are the functional requirements of wearable electronics?
GROOT: “I am Groot.” (They all have similar functional requirements. First, the design and form factor must of the electronic must be unobtrusive and nearly invisible. Second, the electronic components must be reliable to withstand the human environment and last the life of a product. And third, the electronics that come in contact with skin must have a comfortable thermal operating range.
Common applications include vital sign monitoring, safety monitoring, in-vivo implants, chronic disease monitoring, assessment of treatment efficacy systems, and early detection products, to name a few. At the present time, more than 70% of wearable medical devices is for monitoring and feedback.
Many different discoveries and developments enabled the progress of medical wearable devices (MWDs) and are mainly related to advances made in the field of engineering. The major breakthroughs are related to:
- Sensors coupled to microelectronics, telecommunication, and data analysis
- Battery and energy-harvesting technologies
- Telemedicine
- Smartphones, apps, and cloud services
- Communication platforms
As medical wearable devices development progresses, more of the above breakthroughs will appear in production devices and products.)
DANNY: Steve, talk to us about glass core technology and how it can be used in wearable devices?
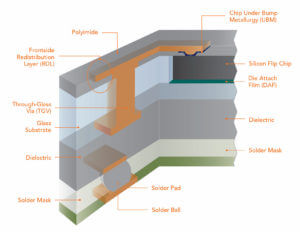
GROOT: “I am Groot.” (GCT is a proprietary process that leverages the performance benefits of glass by creating small diameter, fine pitch Through-Glass Vias (TGV) that are metalized and hermetically sealed. The TGVs are linked via a thin film Redistribution Layer (RDL) process to create custom circuits on a glass substrate. This provides for low loss fan-out of chip and package interconnects, and lower cost compared to traditional silicon-based interposers.
Glass Core Technology has at its basis the capability to provide engineering solutions for connectivity, biomedical, MEMS & Sensors, Optics & Photonics, and hermetic packaging platforms. Glass Core Technology uses standard borosilicate glass, fused silica, quartz, sapphire, and zirconia as the base substrate material.
With the additional features of microelectrode arrays and microneedles, periodic sampling of the wearer’s sweat or blood can provide the needed medical diagnostics and assessments.
Miniaturized components and sensors, advanced power management, and high-performance, low-cost display technologies are making the wearable health market more advantageous for OEMs in the medical devices, and consumer electronics sector. Glass Core Technology provides a much more aggressive set of interconnects than conventional substrates and accelerates the design miniaturization process.)
DANNY: Steve, what is the future direction of medical wearable devices?
GROOT: “I am Groot.” (Since smartwatch manufacturers are leading initiatives for medical wearable devices, I believe the following statement demonstrates the excitement of new technologies: “I don’t look at [smart] watches for their relationship to popular culture, which I know is so much of the fun – but rather as somehow the distillation of craft, ingenuity, miniaturization, and of the art of making.” – Jony Ive)
DANNY: Steve, all of your answers are starting to sound the same to me. Moving forward, can you please elaborate a little more, maybe use more words in your answers?
GROOT: “I am Groot.”
DANNY: Uh, thanks, that’s much better …
You may also be interested in this video / blog — GCT For Miniature Substrate Packaging.