Dissimilar Metals And The Risk of Galvanic Corrosion in Mating Connectors
I am fascinated by the concept of dissimilar metals and galvanic corrosion.
I stayed up until 3:45 am last night surfing the web to learn more. Of course I’m lying, but we do field questions about galvanic corrosion. Robbie Huffman, Samtec’s Interconnect Processing Engineer, recently shared some information on this subject with our Field Sales Engineers. I hope you find it helpful:
When choosing connectors for your application, it is important to choose mating connectors with similar plating materials in the contact areas. Dissimilar metals in the contact areas can result in galvanic corrosion. Galvanic corrosion is an electrochemical process in which one metal corrodes preferentially to another when both metals are in electrical contact, in the presence of an electrolyte.
The chart below shows how different plating materials react to one another with regard to their galvanic potential. The larger the absolute value of the number at the intersection of the two plating materials, the larger the potential for galvanic corrosion. If the number is highlighted in green, that combination of materials has a low galvanic potential and is preferred. If the number is highlighted in yellow, the combination is less than optimal but still acceptable. If it is not highlighted, the potential is high and you may not want to use this combination.
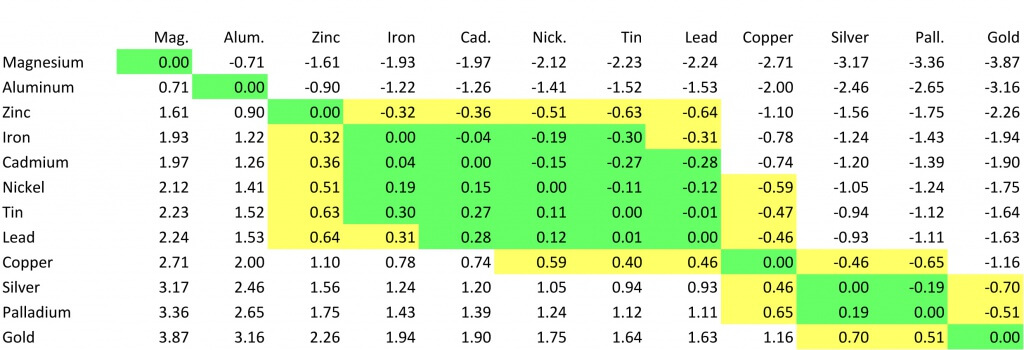
Plating Options On Connectors:
Most PCB level connectors are either gold-plated, tin-plated, or have selective gold/tin. Designers frequently ask what plating finish we recommend. There are numerous considerations to take into account (as evidenced by the variety of plating options on most basic connectors), but the best plating finish is whatever meets your system requirements, at the lowest cost. In other words, make sure it works and meets your quality design specifications, but don’t over-design the plating.
Gold is generally specified for high reliability, low voltage, or low current applications. Gold is used in high cycle applications because it’s rugged and has excellent wear properties (here’s example of a high-cycle connector).
Tin is a lower cost alternative than gold, and has excellent solderability. Unlike gold, tin is not a noble metal. Tin plating starts to oxidize the moment it’s exposed to air. So a tin-plated contact system requires greater normal forces and a longer contact wipe area to break through this oxide film. Hopefully this video shows you what I’m trying to explain. The connector shown is the SSW series.
